The Reticular Activating System
Why every neurologist should know about the RAS and how to examine it.
Case study
A 64-year-old woman with a history of hypertension was found unconscious by her partner. She was well and fully conscious 20 minutes earlier. She was afebrile, with a blood pressure of 164/92mmHg, a heart rate of 87 bpm. On exam, she had asymmetric pupils and withdrawal response to pain in the left arm and leg. There was no response to pain in the right arm and a weak response in the right leg. CT of the brain without contrast did not reveal any acute intracranial abnormality. CT angiography excluded dissection, stenosis, occlusion, or aneurysm. Urgent MRI showed bilateral paramedian thalamic restricted diffusion and bilateral medial thalamic infarction.
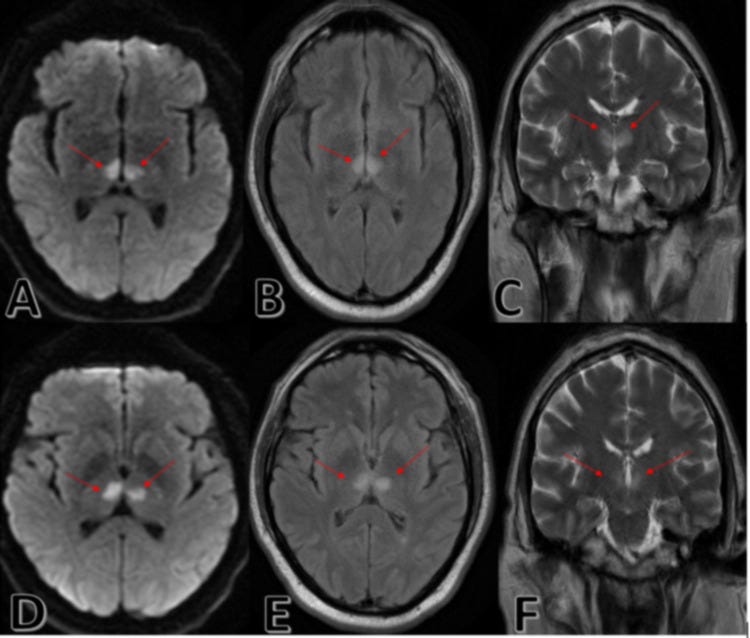
These images from a different patient demonstrate similar changes to those seen in this case. (A, D) Axial DWI MRI view, showing bilateral paramedian thalamic restricted diffusion. (B, E) Axial T2 FLAIR MRI showing bilateral thalamic restricted diffusion in a particular arterial territory. (C, F) Coronal section T2 FLAIR sequence showing bilateral thalamic infarct. (DWI: Diffusion-weighted imaging; FLAIR: Fluid-attenuated inversion recovery; MRI: Magnetic resonance imaging. Images from Wilson et al. Cureus. 2021 Jun 11;13(6):e15587.)
What is the diagnosis?
Bilateral paramedian thalamic infarcts due to an occlusion of the artery of PercheronWhy is the patient unconscious?
Disconnects the reticular activating system arousal inputs via the medial thalamic nuclei that project to the cortex.What system is responsible for maintaining consciousness?
Reticular activating systemHow do you examine this system?
See below
The Reticular Activating System
I recently had to give a lecture on the brainstem to year 1 BSc students and realised that I neglected to spend much time discussing the reticular activating system (RAS). I mentioned it in passing, and, on reflection, this was mirrored in my experience as a medical student and a neurology trainee. Why do we not spend more time discussing, examining and thinking about the RAS when it is the neural substrate that probably underpins consciousness (as defined by neurologists)?
The RAS receives little detailed attention in neurology training. We readily teach cortical localisation, basal ganglia circuitry, and cerebellar ataxia syndromes, but the RAS, which enables all of it to be experienced, is often reduced to a footnote about the brainstem. I will try to close this gap.
The RAS matters clinically because it is critically vulnerable. A discrete lesion in the right location — a few cubic millimetres of tegmentum — can extinguish observable consciousness entirely. Understanding the RAS is therefore inseparable from understanding coma, impaired arousal, sleep–wake disorders, and a range of disorders of consciousness (DoC) that neurologists encounter across the spectrum from acute neurology to neurorehabilitation.
‘A lesion must be either bilateral and hemispheric, or in the brainstem at or above the mid-pons, to abolish consciousness.’
Anatomy and Functional Organisation
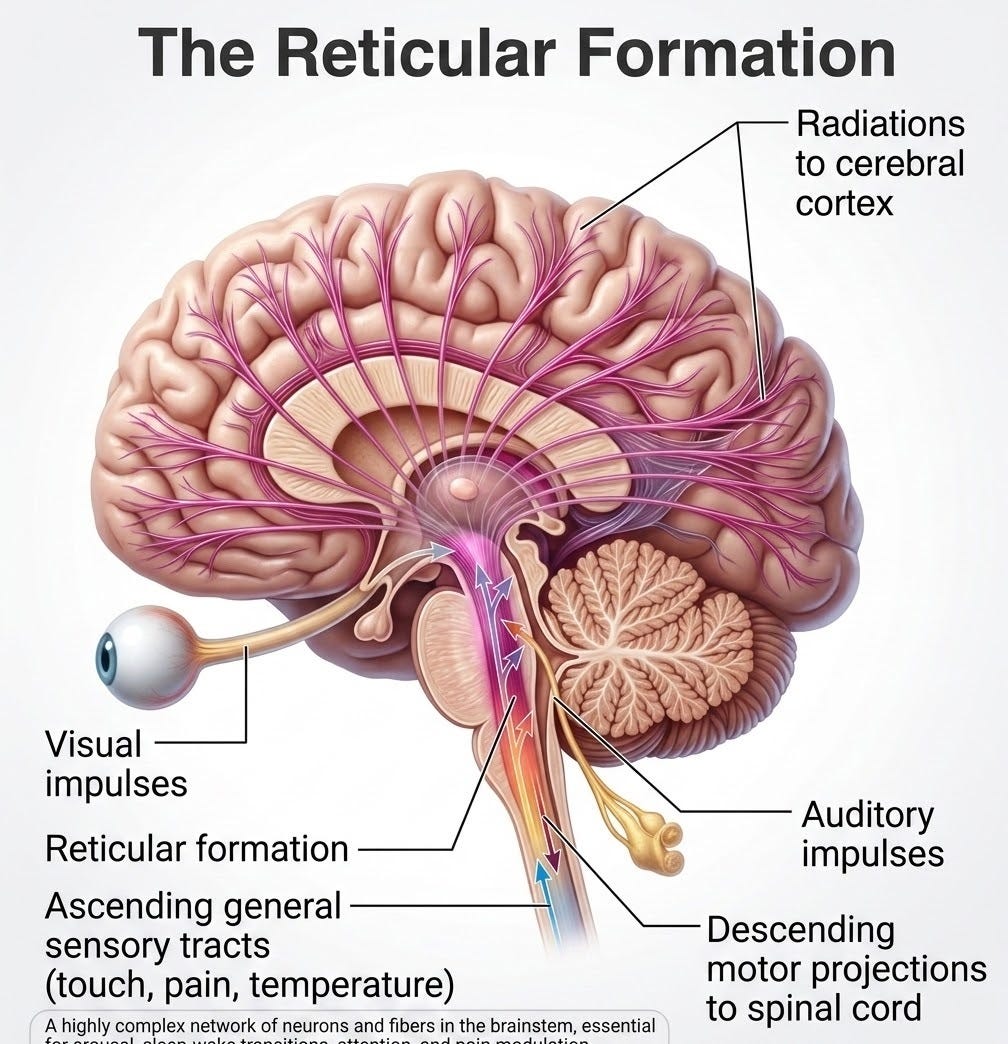
The RAS is not a discrete nucleus but a distributed network of neuronal populations concentrated within the tegmentum of the upper brainstem — specifically the midbrain and rostral pons — with ascending projections to the thalamus, hypothalamus, basal forebrain, and ultimately the cerebral cortex. The key functional components are:
• Cholinergic core: The cholinergic nuclei of the pontomesencephalic tegmentum — the pedunculopontine nucleus (PPN) and laterodorsal tegmental nucleus (LDT). These projects heavily to the thalamus, driving thalamic relay nuclei into the depolarised ‘transmission mode’ necessary for corticothalamic communication. PPN/LDT activity is maximal during wakefulness and REM sleep, and is suppressed during NREM sleep.
• Locus coeruleus: The noradrenergic locus coeruleus (LC), whose diffuse projections across the cortex, hippocampus, and cerebellum modulate arousal, attention, and cognitive flexibility. LC firing is highest during alert wakefulness and is virtually silent during REM sleep.
• Raphe nuclei: The serotonergic dorsal and median raphe nuclei, which project broadly to the forebrain and play a complementary role in arousal, mood, and the gating of sensory information. Like the LC, raphe neurons fire tonically during wakefulness and progressively less during NREM and REM sleep.
• Tuberomammillary nucleus: The histaminergic tuberomammillary nucleus (TMN) of the posterior hypothalamus, whose output maintains wakefulness and whose inhibition by the hypothalamic sleep-promoting ventrolateral preoptic (VLPO) nucleus underlies the sleep–wake flip-flop switch described by Saper and colleagues.
• Orexin system: The orexinergic (hypocretin) neurons of the lateral hypothalamus, which stabilise the flip-flop switch and are critical to sustained wakefulness. Loss of these neurons is the primary pathology of narcolepsy type 1, providing a compelling human disease model of RAS dysfunction.
• Thalamic relay: Relay through the intralaminar and midline thalamic nuclei, which serve as the principal thalamic interface between the brainstem RAS and the cortex. These nuclei project to widespread cortical areas and are required for thalamic desynchronisation — the low-voltage, fast activity on EEG — characteristic of the alert, awake state.
Two ascending pathways carry RAS output rostrally: a dorsal route through the thalamus (the thalamocortical pathway) and a ventral route that bypasses the thalamus to reach the hypothalamus and basal forebrain directly. The existence of both pathways explains why isolated thalamic lesions — while capable of causing hypersomnia or impaired consciousness — rarely produce the complete, sustained coma that tegmental lesions can.
Functional Roles: Beyond Simple Arousal
The RAS is often described as an ‘arousal system’, but this undersells its functional scope. It is more accurate to conceptualise it as a modulator of cortical state — governing the gain, synchronisation, and responsiveness of cortical networks in a context-dependent manner. Its principal functions include:
1. Consciousness and Arousal
The RAS is the necessary, though not sufficient, condition for consciousness. It provides the tonic ascending drive that maintains the cortex in an activated, desynchronised state capable of integrating information. Without intact RAS output, even a structurally intact cortex cannot generate conscious experience — the vegetative state (now termed unresponsive wakefulness syndrome, UWS) illustrates this dissociation, where cortical preservation with disrupted RAS-thalamocortical connectivity results in apparent wakefulness without behavioural evidence of awareness.
2. Sleep–Wake Regulation
The RAS is central to the circadian and homeostatic regulation of sleep and wakefulness. The mutual inhibition between the wake-promoting monoaminergic and orexinergic populations and the sleep-promoting VLPO nucleus generates the bistable flip-flop switch that ensures rapid, complete transitions between sleep and wakefulness. Degradation of this switch — for example, through loss of orexin neurons in narcolepsy, or through diffuse small-vessel disease that disrupts the switch’s stabilising inputs — results in the intrusion of sleep states into wakefulness (cataplexy, hypnagogic hallucinations, sleep paralysis) or in fragmented, unstable sleep.
3. Attention and Cognitive Gating
Noradrenergic LC and cholinergic basal forebrain output modulate cortical attention networks. The LC, in particular, operates according to an inverted-U function of arousal and performance (the Yerkes-Dodson curve), with both hypo- and hyperactivation impairing attentional function. This has direct clinical relevance in traumatic brain injury, where diffuse axonal injury disrupting RAS inputs to the frontal cortex produces the attention and executive dysfunction characteristic of post-concussive and post-TBI cognitive impairment, even in the absence of discrete cortical lesions.
4. Motor System Modulation
The PPN, in particular, plays a prominent role in locomotion initiation and gait and is directly connected to the basal ganglia output nuclei. PPN dysfunction — increasingly recognised as a component of advanced Parkinson’s disease — contributes to falls, freezing of gait, and the postural instability that is poorly responsive to dopaminergic therapy. This has motivated the use of PPN deep-brain stimulation (DBS) as a therapeutic option for selected patients with refractory gait disturbance.
5. Nociception and Autonomic Modulation
Descending reticulospinal pathways from the medullary reticular formation modulate spinal nociceptive processing and sympathetic outflow. The periaqueductal grey (PAG) — a functionally critical component of the descending pain modulatory system — operates in intimate connection with pontine reticular structures. Understanding this connectivity is relevant to the neurological assessment of patients with disorders of consciousness, where preserved nociceptive reflexes may coexist with absent cortical pain processing.
Clinical Consequences of RAS Damage
The clinical signature of RAS dysfunction depends critically on the site, extent, and acuity of damage. A practical framework organises consequences by anatomical level:
Midbrain Tegmentum
Bilateral midbrain tegmental lesions — haemorrhage, infarction, herniation, or demyelination at this level — produce coma with a characteristic oculomotor signature: eyes may be at midposition (loss of both convergence and divergence tone), and the pupillary light reflex is typically absent due to involvement of the Edinger-Westphal nucleus and its outflow. Akinetic mutism can result from less severe bilateral midbrain injury, in which arousal is partially preserved, but voluntary action and speech are abolished. This syndrome is particularly instructive because it reveals the RAS’s role not just in consciousness per se, but in the motivated initiation of behaviour.
Pontine Tegmentum
Rostral pontine tegmental lesions disrupt the PPN/LDT cholinergic input to the thalamus and can cause hypersomnia or coma depending on severity. Central pontine myelinolysis (osmotic demyelination syndrome) characteristically involves the basis pontis with relative tegmental sparing, which is why it produces locked-in syndrome rather than coma — an important anatomical distinction with direct clinical implications. Conversely, haemorrhagic lesions of the pontine tegmentum (often from cavernous malformations or basilar perforator stroke) can be catastrophic because of the density of RAS components at this level.
Thalamic Lesions
Bilateral paramedian thalamic infarction — typically from occlusion of the artery of Percheron, a common trunk supplying both paramedian thalami from the P1 segment — produces a classic syndrome of acute hypersomnia, vertical gaze palsy (due to involvement of the rostral interstitial nucleus of the MLF), and amnesia. Unilateral paramedian thalamic infarction can also cause significant hypersomnia and attentional impairment, reflecting the dominant role of the intralaminar nuclei in thalamocortical arousal. In the chronic setting, thalamic dementia from bilateral thalamic involvement in prion disease (fatal familial insomnia) or Wernicke’s encephalopathy illustrates the devastating cognitive consequences of diencephalic RAS disruption.
Diffuse and Multifocal Dysfunction
Metabolic encephalopathy, sepsis-associated encephalopathy, and toxic states impair RAS function diffusely — typically affecting the metabolically vulnerable monoaminergic nuclei and their projections before causing gross structural damage. This explains why impaired arousal and attentional fluctuation precede coma in most systemic encephalopathies, and why EEG shows progressive slowing (triphasic waves, delta activity) reflecting loss of thalamic desynchronisation. Diffuse axonal injury in TBI similarly disrupts the white matter tracts connecting the RAS to the thalamus and cortex, producing the prolonged disorders of consciousness that characterise moderate-to-severe TBI.
Key clinical principle: In any patient with unexplained impairment of consciousness or arousal, first ask whether the lesion is bilateral and cortical, or in the diencephalon or upper brainstem. The answer determines the differential diagnosis, the imaging strategy, and the prognosis.
Clinical Assessment of the RAS
A structured assessment of RAS function should be part of every neurological evaluation of the unconscious or obtunded patient. The following framework operationalises RAS assessment at the bedside:
Level of Consciousness
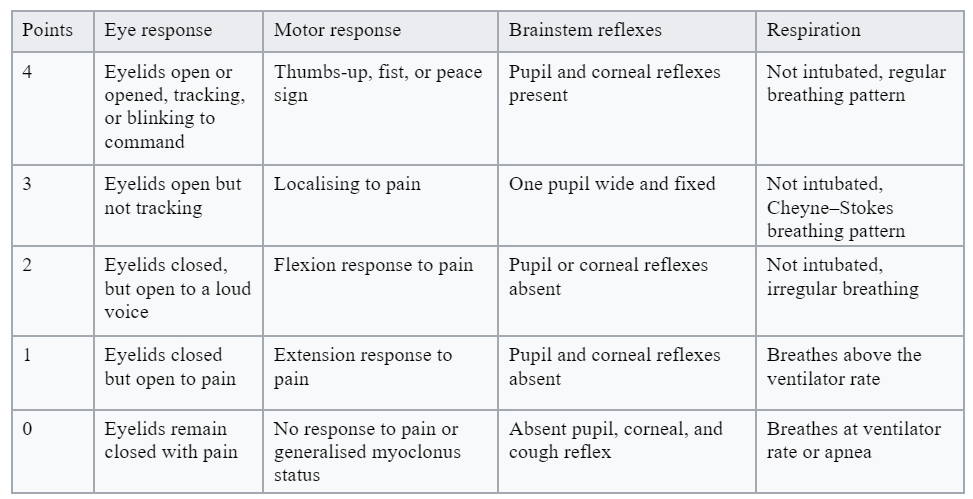
Standardised scales remain the foundation. The Glasgow Coma Scale (GCS), despite its limitations, provides a reproducible index of eye opening (thalamic arousal drive), verbal output (cortical integration), and motor response (corticospinal integrity). The Full Outline of UnResponsiveness (FOUR) scale improves upon the GCS by including brainstem reflexes and respiratory pattern, and is preferable in intubated patients. For patients with chronic DoC, the Coma Recovery Scale-Revised (CRS-R) provides a more sensitive assessment of covert awareness and should be administered by trained staff — repeated measurements increase the detection of command following that may be missed on a single assessment.
Brainstem Reflex Examination
Systematic brainstem reflex testing maps RAS integrity across levels. The pupillary light reflex (afferent CN II, efferent CN III — midbrain level), corneal reflex (afferent CN V, efferent CN VII — pontine level), oculocephalic (doll’s eye) and cold caloric vestibulo-ocular reflexes (pontine-midbrain level), gag reflex (medullary level) and spinocillary (cervical/thoraco-midbrain) together constitute a functional survey of the brainstem from rostral to caudal. Progression of reflex loss in a rostral-to-caudal sequence is the clinical hallmark of transtentorial herniation and warrants immediate intervention.
Spontaneous Eye Movements
Conjugate roving eye movements indicate at least partial brainstem integrity. Dysconjugate gaze or fixed eye deviation points to a focal structural lesion. Ocular bobbing (fast downward, slow return) is virtually pathognomonic of intrinsic pontine pathology. Ping-pong gaze (horizontal alternating deviation every few seconds) typically reflects bilateral hemispheric or diencephalic dysfunction with intact brainstem. These eye movement patterns are directly interpretable through knowledge of RAS anatomy and deserve careful documentation in comatose patients.
Respiratory Pattern
Respiratory pattern is an underutilised indicator of brainstem level. Cheyne-Stokes respiration (periodic crescendo-decrescendo breathing with apnoeas) reflects diencephalic or bilateral hemispheric dysfunction. Central neurogenic hyperventilation, when genuine, indicates midbrain tegmental involvement. Apneustic breathing (prolonged inspiratory pause) points to pontine lesions. Ataxic (Biot’s) breathing is an ominous sign of medullary dysfunction. These patterns are frequently distorted by concurrent metabolic abnormalities and mechanical ventilation, but their presence in a spontaneously breathing patient is localising.
EEG and Neurophysiology
EEG is an indispensable tool for RAS assessment and is underused in general neurology practice. The awake EEG reflects thalamocortical synchronisation driven by the RAS: alpha activity (8–13 Hz) posteriorly during relaxed wakefulness, attenuating to beta with attention. In impaired arousal, progressive EEG slowing — theta then delta — reflects failing RAS drive. Burst suppression indicates severe global cortical dysfunction. The detection of non-convulsive status epilepticus, a common and frequently missed cause of impaired consciousness, requires an EEG. In chronic DoC, EEG-based command-following paradigms and event-related potentials (P300, mismatch negativity) can unmask covert awareness—the electrophysiological counterpart of the clinical CRS-R (coma recovery scale R).
Neuroimaging
MRI is superior to CT for RAS assessment. DWI identifies acute ischaemia in the brainstem tegmentum, thalami, and periventricular white matter — all critical RAS components. FLAIR sequences detect demyelinating lesions in the tegmentum that may otherwise be cryptic, explaining hypersomnia. In TBI, susceptibility-weighted imaging (SWI) demonstrates haemorrhagic diffuse axonal injury along the RAS tracts (dorsolateral midbrain, corpus callosum, grey-white junction). Resting-state fMRI and connectivity analyses are emerging tools for measuring RAS-thalamocortical network integrity in chronic DoC and may increasingly guide prognosis and rehabilitation planning.
Autonomic Assessment
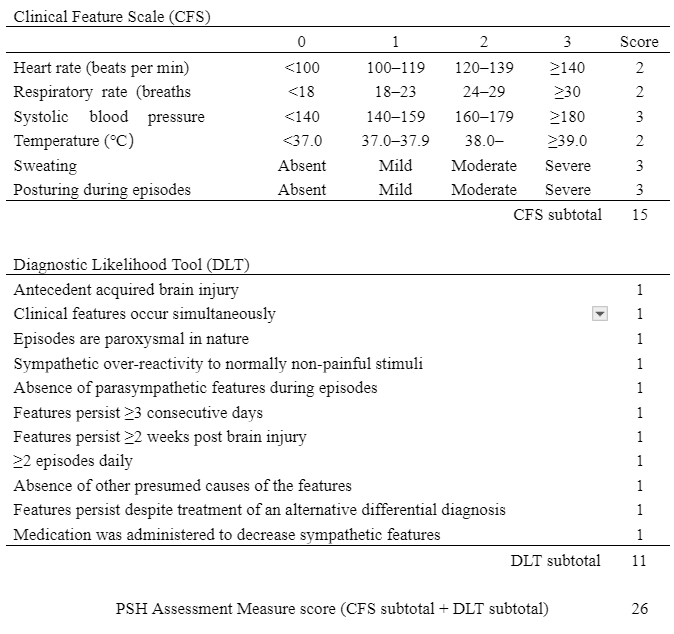
Paroxysmal sympathetic hyperactivity (PSH) — previously termed sympathetic storming — is a manifestation of disrupted descending reticulospinal inhibition of sympathetic outflow, most commonly after severe TBI or hypoxic-ischaemic injury. Its recognition is clinically important because it is frequently misdiagnosed as seizures or infection. The PSH Assessment Measure (PSH-AM) provides a structured diagnostic framework. Conversely, the autonomic tests used to assess the integrity of reticulospinal sympathetic pathways (Valsalva manoeuvre, orthostatic challenge, QSART) are directly relevant to the evaluation of RAS-adjacent pathology in MSA and other synucleinopathies.
Practical Clinical Pearls
• Horizontal eye movement pathways are pontine; vertical pathways are midbrain. A patient with coma and intact horizontal oculocephalics but absent vertical responses has a midbrain-level problem until proven otherwise.
• The presence of sleep-wake cycling (periods of eye opening alternating with eye closure) does not imply awareness — it indicates partial RAS function (sufficient for arousal) but says nothing about cortical processing. Do not conflate eye-opening with consciousness.
• Orexin (hypocretin-1) measurement in CSF should be part of the workup for unexplained hypersomnia. A level below 110 pg/mL is diagnostic of narcolepsy type 1, but low levels also occur in other RAS-disrupting conditions, including encephalitis, TBI, multiple sclerosis (hypothalamic lesions) and paraneoplastic disease.
• The bilateral paramedian thalamic infarction syndrome is misdiagnosed with alarming frequency. Any patient with acute hypersomnia and a normal CT should have an urgent MRI with DWI. The artery of Percheron is invisible on standard angiography; its infarct territory may be the only finding.
• In prolonged DoC after TBI or cardiac arrest, early prognostication should be tempered by awareness that RAS recovery can be slow and non-linear. The concept of ‘late recovery’ — defined as recovery of command following more than 28 days after injury — is well documented and should inform discussions with families before withdrawal of life-sustaining treatment.
The Bottom Line
The RAS is not an anachronism of 20th-century neuroanatomy. It is the gateway through which all neurological function becomes experienced, and its disruption — whether acute and catastrophic or chronic and subtle — generates some of the most challenging and interesting clinical problems in neurology. A fluent command of RAS anatomy, physiology, and clinical correlates may be the distinguishing mark of the competent neurologist, particularly one who wants to be a teacher. It should be taught with the same rigour as motor pathway anatomy, and assessed with the same precision as any other domain of the neurological examination.
Appendix
Glasgow Coma Scale
The Glasgow Coma Scale (GCS) is a clinical tool used to objectively measure a person’s level of consciousness following brain injury, ranging from 3 (deep coma/death) to 15 (fully awake). It assesses three behaviours—eye-opening (1-4), verbal response (1-5), and motor response (1-6)—with a score of 8 indicating severe brain injury.
GCS Components and Scoring
Eye Opening (E): 4-Spontaneous, 3-To sound, 2-To pressure, 1-None.
Verbal Response (V): 5-Oriented, 4-Confused, 3-Inappropriate words, 2-Incomprehensible sounds, 1-None.
Motor Response (M): 6-Obey commands, 5-Localising pain, 4-Normal flexion (withdrawal), 3-Abnormal flexion (decorticate), 2-Extension (decerebrate), 1-None.
Interpretation
Severe: GCS 3–8 (usually requires intubation).
Moderate: GCS 9–12.
Mild: GCS 13–15.
Key Considerations
Limitations: The scale is less reliable if the patient is intubated (often noted as “T” or “NT” for no test) or sedated.
Reporting: It is recommended to report the total score alongside individual components (e.g., GCS 10 = E3, V3, M4) for better accuracy.
Full Outline of UnResponsiveness (FOUR) scale
MANAGEMENT
Clinical management decisions should not be based solely on the FOUR Score in the acute setting and should be used in conjunction with other clinical information.
Very low FOUR scores (≤4) are more predictive of in-hospital mortality than the lowest GCS (3T).
Improvement in score of >2 is predictive of survival in cardiac arrest (Fugate 2010).
Each point increase in the FOUR Score is associated with decreased mortality and morbidity.
CRITICAL ACTIONS
Abnormalities or a decline in score should prompt reassessment and further evaluation for intracranial pathology.
While the FOUR Score may help identify specific states of impaired consciousness, it is important to clearly define the clinical criteria before making these diagnoses.
Paroxysmal Sympathetic Hyperactivity Assessment Measure
The PSH-AM consists of two separate constructs: Clinical Feature Scale and Diagnostic Likelihood Tool, <8 = PSH unlikely; 8–16 = PSH possible; ≥17 = PSH probable. PSH-AM, paroxysmal sympathetic hyperactivity assessment measure
Further Reading
Saper CB, Scammell TE, Lu J. Hypothalamic regulation of sleep and circadian rhythms. Nature. 2005;437(7063):1257-1263.
Giacino JT, Fins JJ, Laureys S, Schiff ND. Disorders of consciousness after acquired brain injury: the state of the science. Nat Rev Neurol. 2014;10(2):99-114.
Posner JB, Saper CB, Schiff ND, Plum F. Plum and Posner’s Diagnosis and Treatment of Stupor and Coma. 5th ed. Oxford University Press, 2019.
Steriade M, McCormick DA, Sejnowski TJ. Thalamocortical oscillations in the sleeping and aroused brain. Science. 1993;262(5134):679-685.
Schiff ND. Recovery of consciousness after brain injury: a mesocircuit hypothesis. Trends Neurosci. 2010;33(1):1-9.